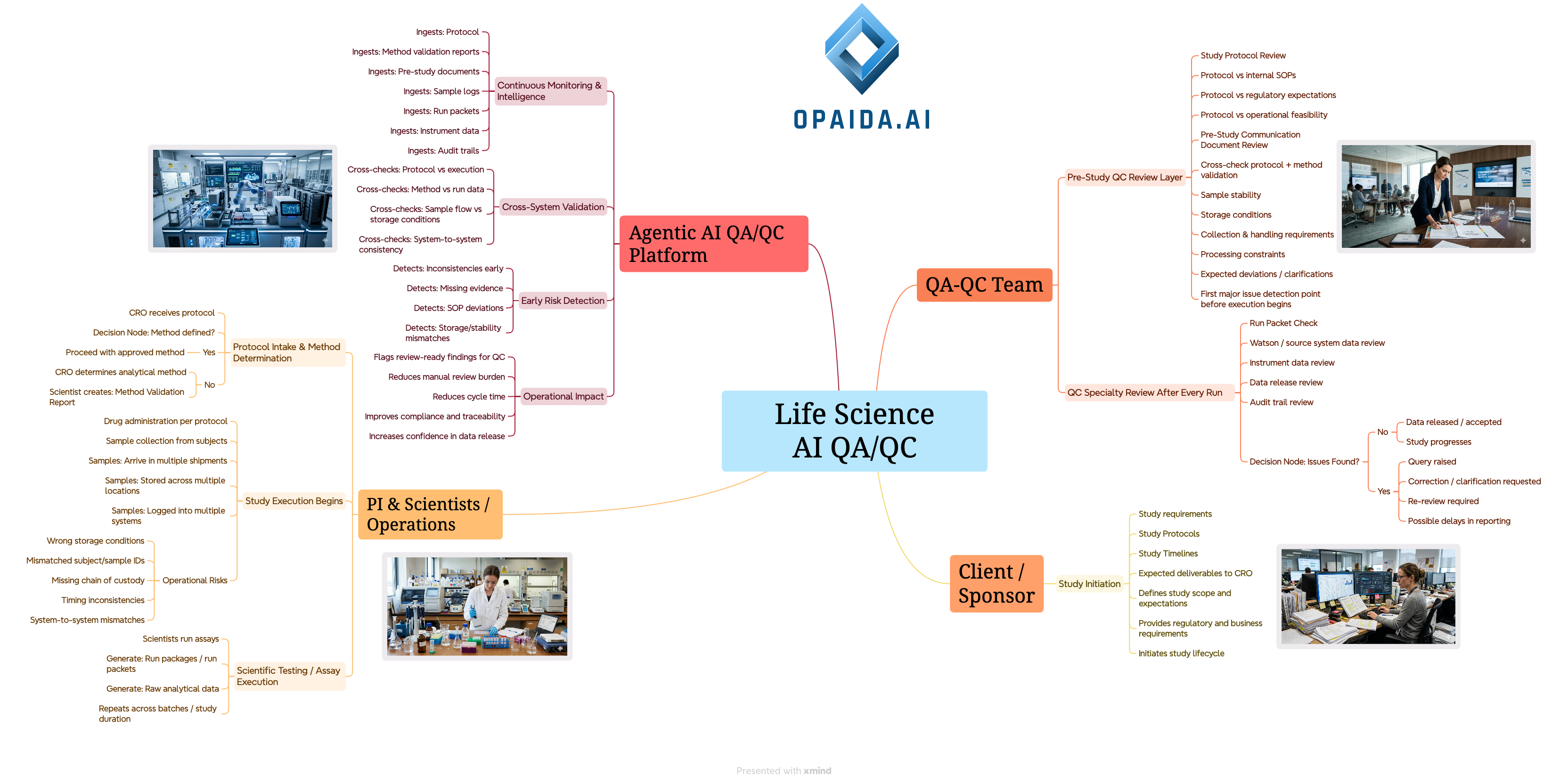
QA/QC & PI
AUTOMATION
Precision Verification & Workflow Acceleration
The Competitive Moat
In the modern life sciences landscape, significantly increasing quality is no longer just a compliance goal—it is one of the single largest competitive advantages. Precision at scale accelerates innovation, reduces time-to-market, and builds unrivaled industry trust.
AI-based QA/QC is the definitive catalyst that brings this strategic advantage to life science companies, transforming manual bottlenecks into high-speed precision engines.
Lost per manual QC cycle
Manual data entry risks
Protocol interpretation issues
Data trapped across systems
Siloed teams & tools
Platform Demonstration
The Solution
Our AI-driven QA/QC and PI automation platform understands protocols and SOPs to eliminate the friction of manual data transfers.
Fragmented data across sub-systems like Watson LIMS, Quickbase, and SharePoint e-Binders is consolidated in real-time, making a comprehensive automated QA/QC checklist possible and more efficient than ever.
Study Binder & Compliance
- • PI/RS Assignment & Training Form Verification
- • Validation Plan & Assay Protocol Alignment
- • Automated Run Packet Completeness Check
E-Notebook (EWB) Review
- • EWB Run ID# vs Watson ID# Reconciliation
- • Method Registration & Signature/Date Audit
- • Analytical Run Status & Review Status Checks
LIMS (Watson) Data Audit
- • Watson Analyte vs MS Peak Area Verification
- • PI-Approved Reassay & ISR Sequence List Validation
- • Comprehensive Study Audit Trail Review
Instrument Data Review
- • Approved Method vs Instrument Method Check
- • Chromatogram Review: Bad Integration Detection
- • IS Evaluation & Carryover Verification
Data Release & Transfer
- • DTA Review & Blinded Data (Alias ID) Verification
- • Predose/Placebo Sample Data Consistency
- • Concentration Unit & Decimal Precision Checks
Archiving & Continuity
- • Archive Index Completeness Audit
- • Knowledge Retrieval for Personnel Continuity
- • Submission-Ready Document Generation
Key Capabilities
-
▶
Cross-System Validation
Automated reconciliation across Watson LIMS, Quickbase, and E-Notebook.
-
▶
Data Entry Automation
AI-driven extraction with zero manual entry and human-in-the-loop review.
-
▶
QA/QC Checklist
Dynamic, automated checklists for protocol compliance and run packet completeness.
-
▶
AI Protocol & SOP Intelligence
Semantic interpretation of complex study protocols, SOPs, and global regulatory frameworks to ensure unwavering compliance.
-
▶
PI Pre-Review Tool
A user-friendly desktop application for Principal Investigators to identify potential issues before formal QA submission.
Workflow Example
Protocol Intake
Client sends requirements & protocol; CRO defines scope
Method Validation
Drug defines method if needed; Validation Report created
Pre-Study QC
Protocol review & communication (stability, storage)
Study Execution
Drug admin & sample collection, shipment & logging
Assays & Analysis
Assays run; run packages created
QC Specialty Review
Run patient data, Watson data, Instruments data, data release review
Drug Development Lifecycle with AI QA/QC
Enterprise-Grade Security
Local Deployment
AI models run entirely on-premise within your own infrastructure — your data never leaves your environment.
Full Audit Trail
Every user operation and system action is tracked and logged, ensuring complete transparency and compliance accountability.
Encryption
Data is encrypted at rest and in transit using industry-standard protocols, protecting sensitive life science data at all times.
Multi-Factor Auth
MFA enforced across all user access points, ensuring only authorized personnel can access the platform and its data.
SYSTEM INSIGHTS
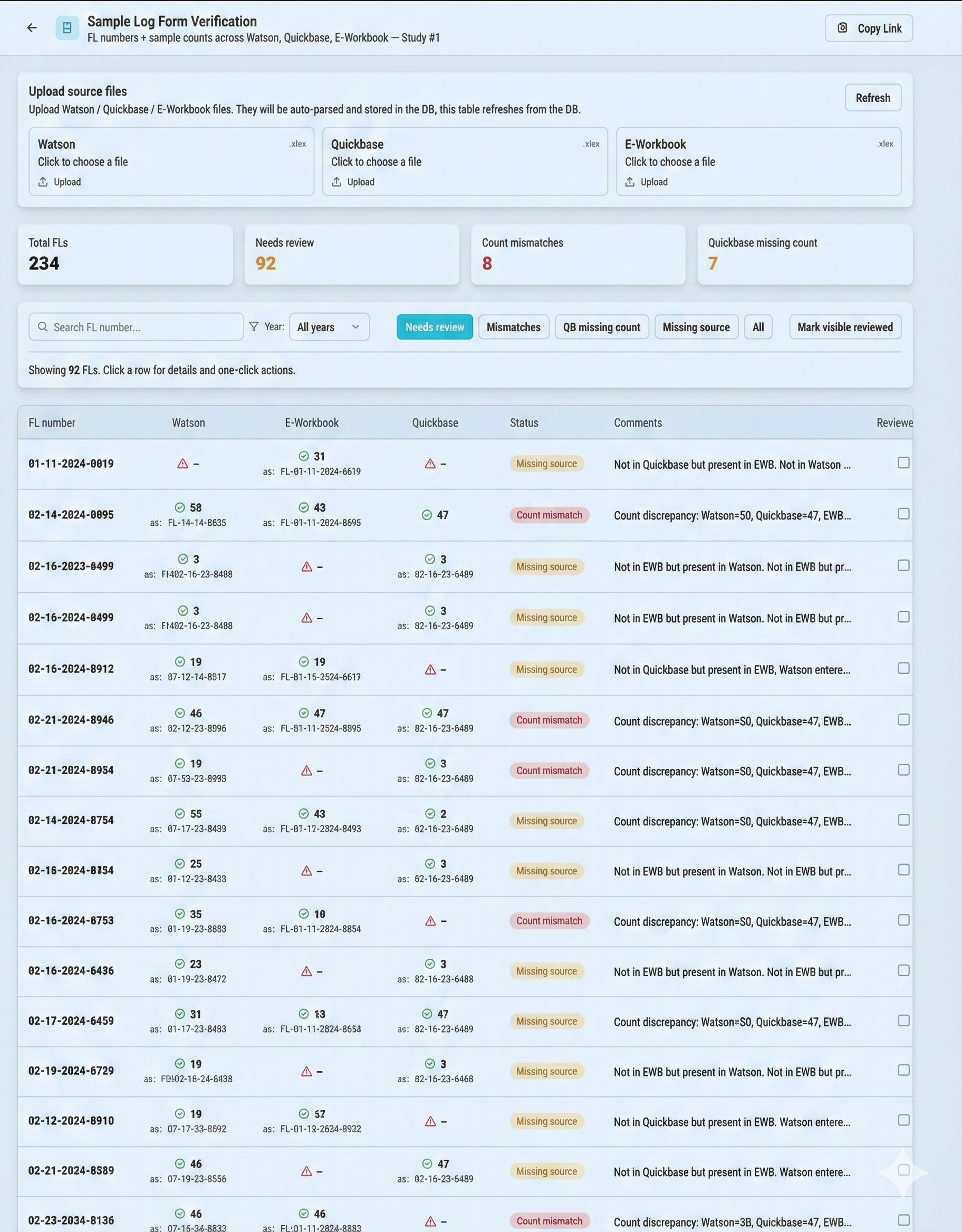
VERIFICATION DASHBOARD
Data Mapping & Ontology

Validation Checklist
SYSTEM INTEGRATIONS
Automated data ingestion via Excel exports for semantic verification and outlier detection.
Real-time synchronization for automated sample tracking, login form verification, and data reconciliation.
Direct live connection to electronic binders for comprehensive document review and cross-system validation.
Seamless extraction and validation of experiment data organized into multi-tab structured formats.